METALS: EXTRACTION of IRON
Iron, the second most abundant metal in the Earth's crust (5.6%), is
usually found as an oxide (e.g., Fe2O3 and Fe3O4 in the ores haematite
and magnetite, respectively). This element, which has been shown to be
essential to all biological species, is a typical transition metal.
Thus, iron has a high melting point (1535°C) and a high density
(7.87 g cm-ł), forms coloured compounds (which are often pale-green or
brown), and shows variable oxidation states [e.g., Fe(II) and Fe(III)].
Furthermore, iron and many of its compounds show catalytic activity
(e.g., iron powder is used in the Haber synthesis of ammonia, and
several iron-containing enzymes are involved in nitrogen-fixation, in
photosynthesis, and in respiration).
[.. K > Ca > Na > Mg > Al > Zn > Fe > Sn > Pb > (H) > Cu > Hg > Ag ..]
1. Iron is extracted from its ores by the chemical reduction of iron
oxides with carbon in a furnace at a temperature of about 800°C;
overall, the processes can be summarized by these equations:
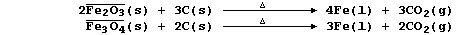
Coke, the source of chemical energy in the blast furnace, is burnt both
to release heat energy and to provide the main reducing agent:

Calcium oxide, formed by thermal decomposition of limestone, reacts
with the silicon oxide present in sand, a major impurity in iron ores,
to form slag (which is less dense than molten iron):
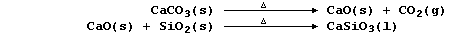
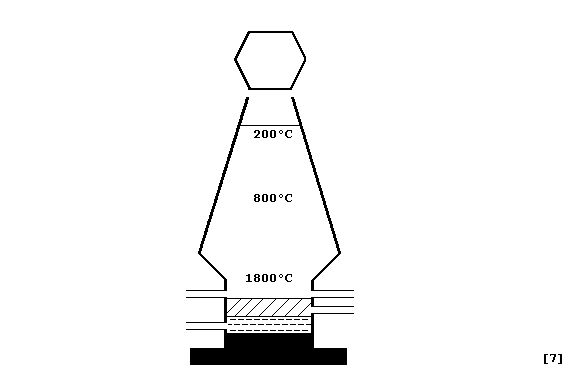
(a) Label this diagram of an iron blast furnace with: Blast of hot air;
Molten iron; Molten slag; Outlet for iron; Outlet for slag; Reactants
(iron ore, coke, and limestone); and, Waste gas outlet.
(b) Various iron oxides are formed within the furnace. Construct the
symbol equation for the reduction of iron(II) oxide by carbon monoxide.
_______________________________________________________________________
[2]
(c) Name two of the waste gases. ______________________________________
[2]
2. Nearly all metals corrode: but only iron 'rusts'. The rusting of
iron, which starts with the anodic reaction Fe(s) ——® Fe2+(aq) + 2e-,
involves a complicated series of redox and precipitation reactions that
can be loosely summarized by the following equation (where, n and x are
variable numbers, and z and y are constants):
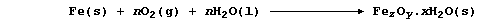
(a) Laboratory analysis showed that a sample of rust contained 5.6 g of
iron, 2.4 g of oxygen, and 2.7 g of water. The steps involved in this
analysis included strong heating of the hydrated oxide, to remove its
water of crystallization, followed by strong heating of the anhydrous
oxide in a stream of dihydrogen, to effect its reduction; i.e.,
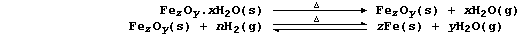
|
Fe |
O |
H2O |
Mass combining (m) / g |
|
|
|
Molar mass (M) / g mol-ą |
|
|
|
Number of moles combining (m ÷ M) |
|
|
|
Simplest ratio of number of moles |
|
|
|
Complete the Table above, so as to determine the empirical formula of
this sample of rust. __________________________________________________
[5]
Rusting occurs faster in the presence of aqueous chloride ions; a
partial explanation for this observation is as follows. Corrosion is an
electrochemical process, in which one part of the metal surface acts as
the anode, another part acts as the cathode, and an aqueous electrolyte
completes the electrical cell. Electron flow from anode to cathode is
facilitated by migration of ions such as Fe2+(aq) in the electrolyte;
and, because pure water is only very slightly ionized, an aqueous
solution of chloride ions is clearly a better electrolyte than water.
(b) State two ions present in 'acid rain' that might also increase the
rate of rusting / corrosion. __________________________________________
[2]
Iron's widespread use as a structural material is due to its low cost
and high tensile strength. Unfortunately, rust weakens structures;
moreover, it cannot protect the metal from further corrosion because it
is porous. Accordingly, apart from important safety considerations, the
costs of possible replacements need to be minimized. And so, when iron
(or steel) is likely to rust, it is: painted, coated with plastic, or
plated with another metal, all of which provide a physical barrier; or
attached to a more reactive metal, which acts as a 'sacrificial anode'.
(c) Zinc is more easily oxidized than iron. Therefore, in an electrical
cell, the preferred anodic reaction Zn(s) ——® Zn2+(aq) + 2e- results in
electron flow to an iron cathode, and so prevents the anodic reaction
Fe(s) ——® Fe2+(aq) + 2e-. Thus zinc acts as a sacrificial anode, and so
blocks of zinc attached to iron structures provide effective protection
against rusting. Explain, in similar terms, the effectiveness of:
Magnesium blocks ______________________________________________________
_______________________________________________________________________
_______________________________________________________________________
Tin blocks ____________________________________________________________
_______________________________________________________________________
[5]
(d) Iron coated with a layer of zinc is known as 'galvanized iron'.
Galvanization protects iron from rusting, as follows: the layer acts as
a physical barrier to dioxygen and water; and then the zinc acts as a
sacrificial anode when this layer is breached. Suggest one disadvantage
in galvanizing iron. __________________________________________________
[1]
Dr. R. Peters Next Contents' List